
And then there are the classics. Catnip might be basic but it’s inexpensive and most cats go crazy for it. There are also more practical items like robotic litter trays and automatic cat flaps.
There are a lot of products to choose from, and to help you find the best products for cats, we have included reviews of the best ones below.
A Quick Comparison of Our Favorites in 2024
| Image | Product | Details | ||
|---|---|---|---|---|
| Best Overall |

|
Hepper Hi-Lo Scratcher |
|
CHECK PRICE |
| Best Value |

|
SmartyKat Silvervine Cat Attractant Catnip |
|
CHECK PRICE |
| Premium Choice |
|
Litter-Robot 4 with Step & Core Accessories |
|
CHECK PRICE |

|
Hepper Stainless Steel Cat Water Fountain |
|
CHECK PRICE | |

|
K&H Pet Products Deluxe Kitty Sill Cat Window Perch |
|
CHECK PRICE |
The 15 Best Products for Cats
1. Hepper Hi-Lo Scratcher – Best Overall

| Product Type: | Scratcher |
| Materials: | Plywood, Corrugated Cardboard |
Cats love to scratch and if you don’t provide an appealing surface that the cat is allowed to scratch, they will choose their own scratching surface. The Hepper Hi-Lo Scratcher helps save furniture, carpet, and doors while offering a choice of three scratching positions: high, low, and lower.
The frame is made from plywood, so it is light but sturdy, and the scratching surface is made of corrugated cardboard. Replacement scratcher pads can be bought separately so you don’t need to replace the scratcher every time your cat gets through a couple of layers. The scratcher is reasonably priced, looks good in the house, and the glossy plywood can be wiped clean, if needed, making it the best overall product for cats this year.
However, some cats prefer the upright cat scratcher design that lets them stretch out and up while using it.
At Catster, we’ve admired Hepper for many years and decided to take a controlling ownership interest, so that we could benefit from the outstanding designs of this cool cat company!
- Choice of scratching positions
- Attractive design will appeal to humans, too
- Cardboard scratching surface can be replaced
- Reasonably priced
- Some cats will prefer the upright tower scratching position
2. SmartyKat Silvervine Cat Attractant Catnip – Best Value

| Product Type: | Catnip |
| Materials: | Catnip |
Approximately 60% of cats are reactive to catnip, and if yours is one of them, you can send them into rapture with a sprinkle of the mint family member 1. While your cat is rubbing themselves against the catnip, it will heighten their senses and make your feline friend more alert, while ingestion of the catnip is safe and will calm your cat. You can just sprinkle some on the ground, or you can use it to attract your cat to the new toy you’ve bought that has garnered no interest.
SmartyKat Silvervine Cat Attractant Catnip is a tub of catnip, with no other ingredients. It’s basic but effective for most cats, and it really can work well to encourage your kitty into new cat beds, scratchers, and other items, or it can make playtime more entertaining. It’s also harmless and inexpensive, making it the best product for cats for the money.
But, if your cat is part of the 40% of the feline population that catnip doesn’t affect, it will prove useless.
- Inexpensive
- 60% of cats are reactive to catnip
- Can be used to encourage your cat to use new beds, toys, etc.
- Not all cats react to catnip the same way
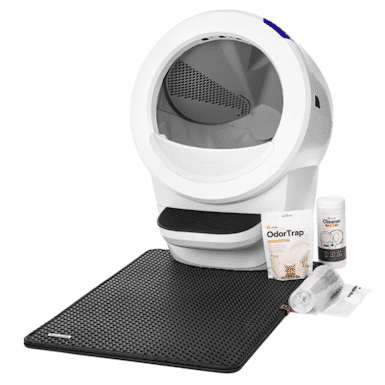
3. Litter-Robot 4 with Step & Core Accessories Kit Cat Litter Box – Premium Choice

| Product Type: | Litter |
| Materials: | Plastic |
Keeping cats means keeping cat litter trays, and that means cleaning them out regularly. Cleaning the litter tray not only prevents the smell from escaping and traveling through the house, but some cats will refuse to use a dirty tray, leading to inappropriate littering outside the tray. But, whether you’re at work all day, have multiple cats that like to use the same tray, or struggle to lean over and keep the tray clean, it can be a bind to keep the tray tidy.
The Litter-Robot 4 with Step & Core Accessories Kit Cat Litter Box is a self-cleaning litter tray. It essentially sifts the litter to get rid of solids and litter clumps. It also comes with litter liners and odor eliminators, and this pack includes a step, a fence, and a litter-trapping mat to help prevent the tracking of litter through the house.
The Whiskers app can let you know when the tray is full and even monitor your cat’s usage so you can determine whether they are peeing or pooping more or less frequently.
- Automatically cleans the litter
- Prevents litter tracking
- Odor eliminators help combat bad smells
- Very expensive
4. Hepper Stainless Steel Cat Water Fountain

| Product Type: | Water Bowl |
| Materials: | Stainless Steel |
Cats are really very intelligent. When they can be bothered. They instinctively know that drinking from stagnant water sources increases the risk of ingesting bacteria and other waterborne nasties. They understand that flowing water is safer to drink, and that’s why a lot of cats refuse to drink directly from a water bowl. And it’s why some cats exhibit the seemingly odd behavior of splashing around in their bowls before drinking. If yours is one of those cats that won’t drink from their water bowl but enjoys lapping at a dripping faucet, a water fountain can be a good solution.
The Stainless Steel Cat Water Fountain is quiet, so it won’t disturb you even when you’re nearby. It has a triple-filtration system that softens tap water and removes impurities, and there is a choice of three water speed settings. It is reasonably priced for a stainless steel fountain and is a good option for those cats that refuse to use a water bowl. However, if your cat likes to play with running water, the fountain will encourage it and could lead to spillage accidents.
- Good for picky drinkers that won’t use a water bowl
- Quiet operation
- Reasonable price for quality materials
- Can encourage cats to play with water and spill
At Catster, we’ve admired Hepper for many years and decided to take a controlling ownership interest so that we could benefit from the outstanding designs of this cool cat company!
5. K&H Pet Products Deluxe Kitty Sill Cat Window Perch

| Product Type: | Cat Bed |
| Materials: | Plastic, Synthetic Fabric, Faux Fleece |
Windows are essentially like TVs for cats. They can watch birds fly by and people and other animals walk past, depending on where they live. They also enjoy sitting by the natural radiators because of the heat the sun gives off. You can clear all the ornaments and leave space for your cat to sit, but better still is providing a comfortable, cushioned perch for your favorite feline to rest on.
The K&H Pet Products Deluxe Kitty Sill Cat Window Perch uses a hook and loop to attach to the sill, which means there’s no need for tools and assembly is easy. The manufacturers claim a maximum weight capacity of 40 pounds which should be ample for even the biggest house cat. And the cover can be removed and is machine washable so it’s easy to care for.
However, the K&H Pet Products Deluxe Kitty Sill Cat Window Perch is quite expensive and despite the weight limit, it will struggle with cats that weigh much more than 20 pounds.
- No tools required for assembly
- Fixes to the window sill
- Cover is machine washable
- Quite expensive
- Only really suitable for cats up to 20 pounds
6. Frisco Hexagon Wall Mounted Cat Wall Shelf

| Product Type: | Cat Bed |
| Materials: | Engineered Wood, Plush |
The Frisco Hexagon Wall Mounted Cat Wall Shelf is a hollow hexagonal-shaped shelf that fixes to the wall and contains a soft pillow for your kitty to lay on. The top of the shelf is also padded and offers an alternative position for cats that don’t want to be enclosed but still want the elevated position.
Cats like elevated positions because they offer security and safety, and because the front of the shelf is open, your cat can still watch what’s going on around the room while enjoying somewhere comfortable to sit. The cushion can be removed and washed. And the shelf is modular, with gaps and holes on each side so you can add tunnels, other shelves, or erect a cat tree to offer vertical access to the shelf.
The shelf is easy enough to construct, but some cats might struggle to climb in, and some won’t appreciate the open sides that can leave them feeling exposed.
- Offers an elevated perching and sleeping position
- Cushion can be removed and washed
- Easy to erect
- Some cats won’t want to climb in
7. Frisco Cat Silhouette with Bridge Wall Mounted Cat Wall Shelves

| Product Type: | Wall Shelves |
| Materials: | Engineered Wood, Polyester |
For those cats that do enjoy scaling heights and climbing walls, you don’t need to stop at a wall-mounted cat bed. The Frisco Silhouette with Bridge Wall Mounted Cat Wall Shelves set fixes to the walls, so you will need to screw it into your walls. It includes two condo shelves that your cat can fit and sit in, with a bridge connecting the two.
You could place one of the boxes next to the cat shelf above to create a vertical living area for your feline friends. The condos have sisal mats on the floor, for scratching, and you get a wand toy that you can use to encourage reluctant cats onto their new playing surfaces.
- Uses wall space, rather than floor space
- Offers an elevated position
- Sisal scratch mats on every shelf
- You do need to screw into the wall
8. Mau Lifestyle Cento 46-In Modern Wooden Cat Tree & Condo

| Product Type: | Cat Tree |
| Materials: | Wood, Faux Fur, Synthetic Fabric |
A cat tree is another product that allows your cats to climb, scratch, and hang out. Trees are tall, have multiple branches, and they tend to incorporate extra features. But they can be quite ugly. The Mau Lifestyle Cento 46-In Modern Wooden Cat Tree & Condo is a 46-inch-tall tree with two main columns.
On top of one column is a nest bed. The other column has a condo-style base and another nest bed on top. One of the trunks has a scratching post and both beds have hanging toys. The columns are made from real tree branches so they are strong and look natural. The beds are made of woven baskets and have machine-washable cushions inside.
Although it is expensive, the Mau Lifestyle Cento 46-in Modern Wooden Cat Tree & Condo is better looking than a lot of alternatives and the use of real branches makes it very sturdy.
- Made of natural materials for an attractive finish
- Scratcher, bed, and hanging toys
- Cushion can be removed and machine-washed
- Expensive
- More activities on other, cheaper trees
9. Hepper Harness and Leash Set

| Product Type: | Leash |
| Materials: | Velvet, Mesh, Velcro, Aluminum |
Keeping your cat indoors can prevent illnesses and other incidents, but cats do love to spend time outdoors. One solution is to fasten on a harness and leash and walk them to your local park or take them on hikes with you. It can take time to get a cat used to wearing a harness or walking on a leash, but it does offer them much greater freedom.
The Hepper Harness and Leash Set has a soft, comfortable harness that is adjustable, so it fits snugly on your cat. It has reflective stripes to ensure that drivers and others can see you and your cat while you’re out, and it comes in a choice of five sizes.
Unfortunately, some cats will likely never really take to being walked on a leash, especially if they’re nervous or anxious around dogs, cats, cars, or people.
- Choice of five sizes to ensure a good fit
- Velcro strap ensures a snug fit
- Reflective stripes improve visibility
- Some cats don’t take to being walked on a leash
At Catster, we’ve admired Hepper for many years and decided to take a controlling ownership interest so that we could benefit from the outstanding designs of this cool cat company!
10. High Tech Pet Products PX-1 Power Pet Fully Automatic Pet Door

| Product Type: | Pet Door |
| Materials: | Plastic |
If your cat does have the freedom of being allowed outdoors, a pet door prevents you from having to open and close the door at all hours.
The High Tech Pet Products PX-1 Power Pet Fully Automatic Pet Door uses an RFID chip, attached to your cat’s collar, that opens the door automatically when your cat approaches. The door slides up into the frame, so there’s no pushing involved, and you can set the door to in-only, out-only, fully locked, or full access. It also has dual range access, so you can set the door to open from different distances depending on whether your cat is coming in or going out. Additional collars can also be purchased so you can have multiple cats use the same door.
The pet door is expensive and while it is beneficial to have the door sliding up and down automatically, it lacks the timer feature of some other cat door models.
- Multiple cats can use the same door
- Opens and closes automatically
- Let your cat have the freedom of the outdoors
- Expensive
- No timer options
11. Furhaven Multi-Level Cardboard Cat Condo

| Product Type: | Condo |
| Materials: | Cardboard |
The Furhaven Multi-Level Cardboard Cat Condo is a 4-story cat condo made from double-walled, corrugated cardboard. The condo does need putting together, but its plain cardboard walls can be decorated inside and out, and the cardboard is thick enough that it will withstand scratching.
The condo includes catnip to entice your feline friends inside, as well as several detachable toys to add extra elements of enjoyment for your cat. It does work out cheaper than buying a pre-assembled 4-story condo toy, but feels expensive for a cardboard toy, and if you have very active, larger cats that like the top layers, it won’t last forever before it starts to topple under the weight.
- Multiple levels for cats to enjoy
- Cardboard is safe to scratch
- Includes catnip and detachable toys
- Does need constructing
- Might topple with large, active cats
12. Cat Scratch Pad Skateboard

| Product Type: | Scratcher |
| Materials: | Corrugated Cardboard |
Cats are inquisitive and playful. While this Cat Scratch Pad Skateboard isn’t a working skateboard – it is made of cardboard after all – it does offer a scratching surface for your cat and a fun design for you. The whole thing is scratchable and because it’s made of corrugated cardboard, it won’t be too easily destroyed. It arrives fully assembled so you don’t need to put it together when it arrives.
The skateboard is reasonably priced but because it is made of cardboard it is quite lightweight so it is easily moved around when your cat scratches it. The movement can put some cats off using it, and you may need to push it up against a wall so your feline friend can get a decent purchase on it.
- Good-looking design
- No need to construct it
- Whole thing is scratchable
- Moves around easily

| Product Type: | Cat Treadmill |
| Materials: | Wood, Felt |
The Coziwow Exercise Wheel with Lock & Teaser Cat Treadmill is a giant exercise wheel suitable for most cats. The use of the wheel is similar to that of a hamster wheel. Your cat gets in it and runs, giving them exercise and allowing them to burn off any pent-up energy.
The Coziwow wheel is quite large and has a removable carpet that can be cleaned and washed to keep it fresh. The wheel also includes a teaser wand that you can use to encourage the cat on the wheel and to start running, effectively teaching your feline friend how to use their new piece of equipment. The wheel is supposed to be able to hold up to 110 pounds, which means it has more than enough capacity for a household cat or two.
The Coziwow Exercise Wheel with Lock & Teaser Cat Treadmill is expensive and it is very challenging to put it together when it arrives.
- Provides plenty of daily exercise
- Includes a wand toy to encourage wheel use
- Carpet can be removed and washed
- Expensive
- Tricky to put up
14. PetAmi Deluxe Backpack Dog & Cat Carrier

| Product Type: | Backpack |
| Materials: | Synthetic Fabric, Polyester |
For owners that want to take their cats out hiking, regularly travel with them, or want an alternative to a cat carrier for getting to and from the vet and other locations, a cat backpack is one alternative.
The PetAmi Deluxe Backpack Dog & Cat Carrier can carry up to 18 pounds and won’t be suitable for Maine Coons and other giant breeds but should be big enough for most cats. It has ventilated mesh panels that allow air in and out. It has a reinforced frame to prevent the bag sides and top folding in on your cat during transit, and it has a waist strap as well as chest straps to disperse the load around your body during transit.
However, for cats at or near the weight limit, space will be very restricted, and the back of the backpack would benefit from being padded to offer greater comfort for the wearer.
- Provides plenty of daily exercise
- Includes a wand toy to encourage wheel use
- Carpet can be removed and washed
- Expensive
- Tricky to put up
15. Shele UFO Interactive Cat Toy

| Product Type: | Interactive Toy |
| Materials: | Plastic |
Some cats can entertain themselves for hours with toys or even household objects they’ve purloined from their owners. Others prefer interactive toys that encourage them to play, but even if you spend a lot of time with your cats, you may not be able to play whenever your four-legged friend demands it.
A toy like the Shele UFO Interactive Toy means your cat can enjoy interactive games even when you’re not available. The UFO shaped toy has feathers that pop in and out of the base of the toy. It lights up, too, and different modes and settings alter the speed of the feather as well as the lights of the toy. It is rechargeable so you don’t need to keep buying batteries, and it can be set to smart mode so it only operates when it senses your cat’s claws.
However, you may need to supervise play time because if your cat can catch the feathers, the toy is light enough to be dragged around, potentially ruining the motor.
- Interactive toy entertains your cat
- Variable settings to tailor to your cat
- Rechargeable so no need to buy new batteries
- Lightweight and easily pulled around
Buyer’s Guide: Selecting the Best Products for Cats
Cats are inquisitive, playful, and typically energetic little pets. While some are content sleeping in cardboard boxes and playing with small toys or household items, others are more demanding.
Product Types
The best cat products are those that your cat will enjoy using or benefit from. If you have a lively cat that would benefit from some extra exercise, consider toys or a wheel. If your cat loves nothing more than lazing in the sun or watching birds out of the window, get a window bed. There are a lot of different products and items available, with different purposes and properties, so you’re sure to find something to make your feline friend happy and content.
Exercise
Cats need regular exercise, just like dogs do. For those that have free reign outdoors, this shouldn’t be too challenging. But, for indoor cats, or those that don’t necessarily like to venture far from the door, items like exercise wheels and cat trees encourage active play.
Feeding and Watering
Felines also need good daily nutrition and they need to hydrate through the consumption of water. But not all cats like to drink straight from a water bowl. Fountains and other water accessories can help you ensure your cat is getting their daily water intake.

Sleeping and Naptime
Cats can sleep 16 hours a day. They don’t usually sleep for a single 16-hour session and may wake up and wander around for half an hour before going back to their bed or a favorite spot for a power nap. The amount a cat sleeps means it needs these comfortable spots. As such, there are a lot of different beds and bed styles available. Some of the most popular are those that allow your cat to hide away, or those that offer an elevated position from where your cat can survey the surrounding environment.
Interaction and Interactivity
Interacting with your cat helps build a bond and it can also provide the cat with physical exercise and mental stimulation. Certain types of toys encourage this interaction, but you can’t always be at your cat’s beck and call. Interactive toys entertain, amuse, and excite your cat. You can join in with the games or let your cat fly solo.

Carriers and Walkers
Like dogs, you can walk some cats on leashes, but not all cats will take to the wearing of something so constraining. Instead, you can buy backpacks to carry your cat around the park or to their vet appointment.
Conclusion
Cats have a lot of different requirements, from activity and exercise to mental stimulation and interaction. Above are reviews of 15 of the best products for cats to help you care for, entertain, and stimulate your feline friend.
The Hepper Hi-Lo Scratcher is reasonably priced, looks good, and offers your cat several scratching positions to choose from. SmartyKat Silvervine Cat Attractant Catnip is a tub of catnip, which is a favorite of many cats, and also keeps their owners entertained.
Featured Image Credit: Garmasheva Natalia, Shutterstock
Contents
- A Quick Comparison of Our Favorites in 2024
- The 15 Best Products for Cats
- 1. Hepper Hi-Lo Scratcher – Best Overall
- 2. SmartyKat Silvervine Cat Attractant Catnip – Best Value
- 3. Litter-Robot 4 with Step & Core Accessories Kit Cat Litter Box – Premium Choice
- 4. Hepper Stainless Steel Cat Water Fountain
- 5. K&H Pet Products Deluxe Kitty Sill Cat Window Perch
- 6. Frisco Hexagon Wall Mounted Cat Wall Shelf
- 7. Frisco Cat Silhouette with Bridge Wall Mounted Cat Wall Shelves
- 8. Mau Lifestyle Cento 46-In Modern Wooden Cat Tree & Condo
- 9. Hepper Harness and Leash Set
- 10. High Tech Pet Products PX-1 Power Pet Fully Automatic Pet Door
- 11. Furhaven Multi-Level Cardboard Cat Condo
- 12. Cat Scratch Pad Skateboard
- 13. Coziwow Exercise Wheel with Lock & Teaser Cat Treadmill
- 14. PetAmi Deluxe Backpack Dog & Cat Carrier
- 15. Shele UFO Interactive Cat Toy
- Buyer’s Guide: Selecting the Best Products for Cats
- Conclusion













